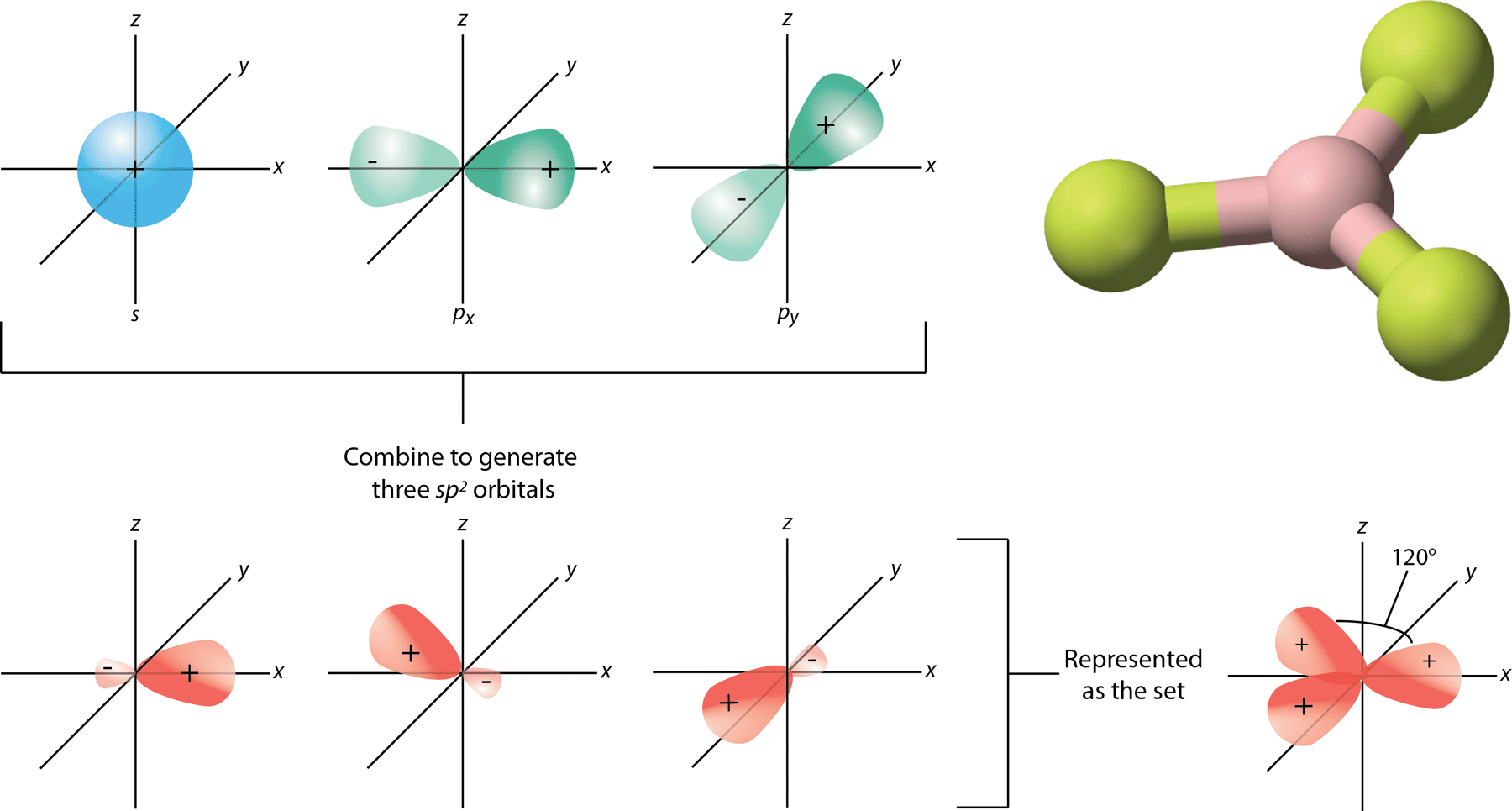
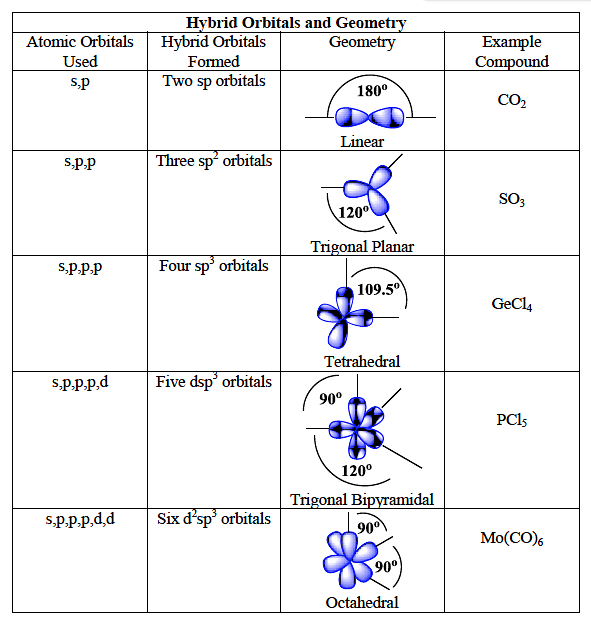
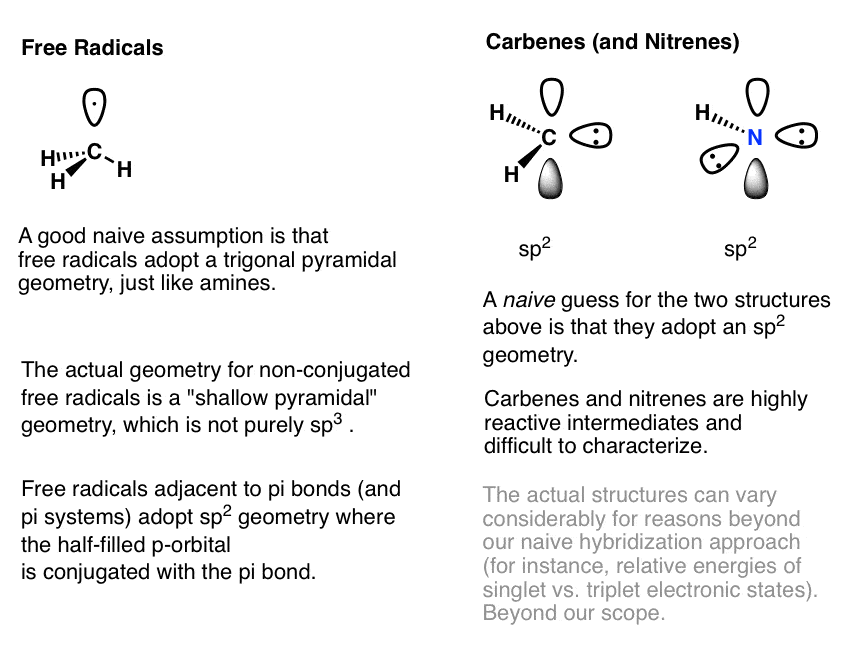
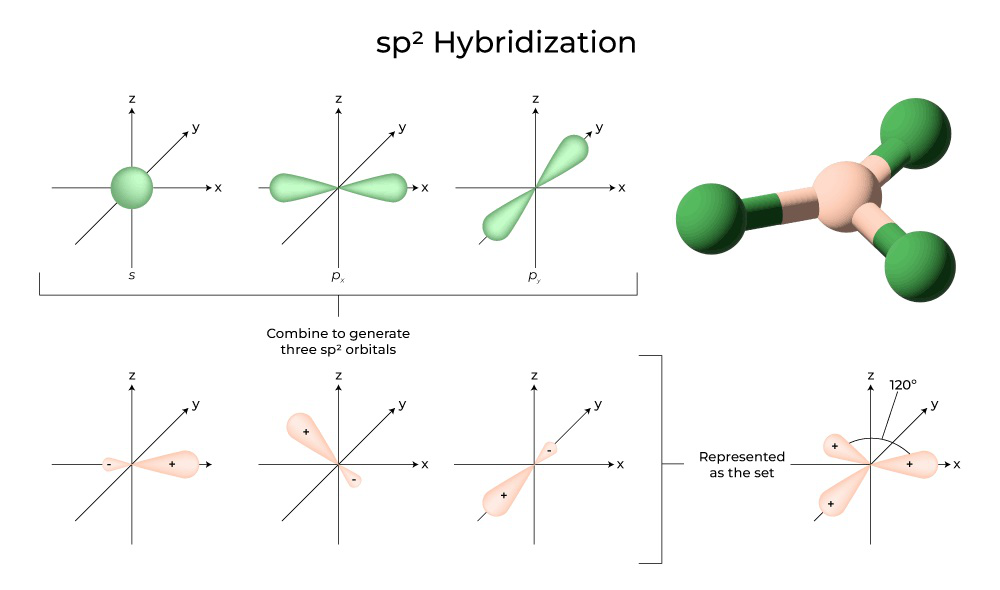
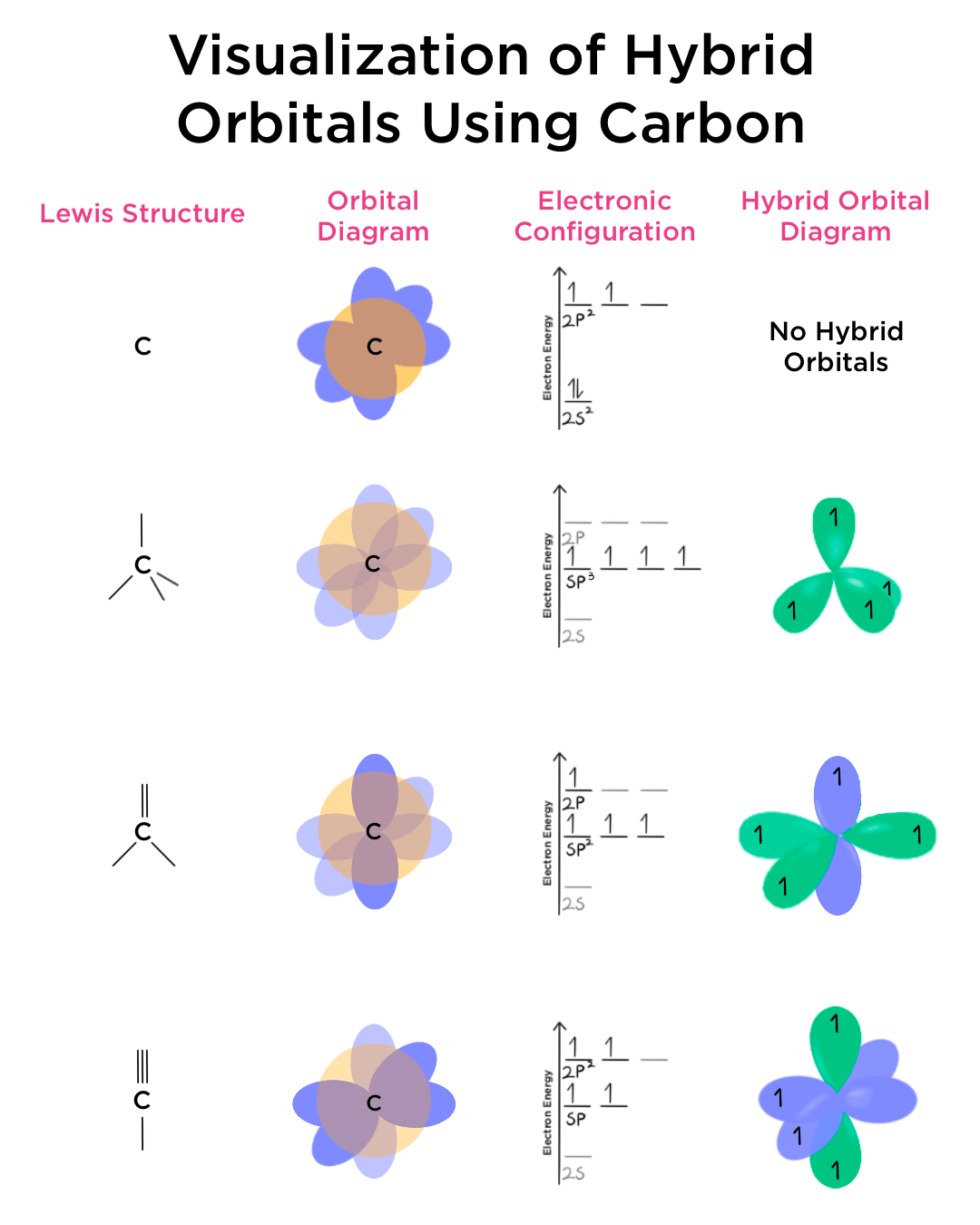
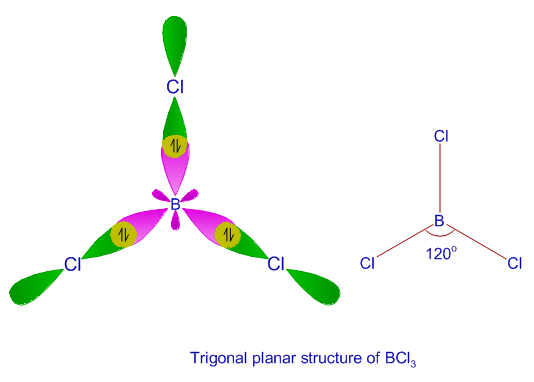
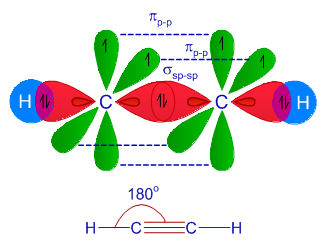
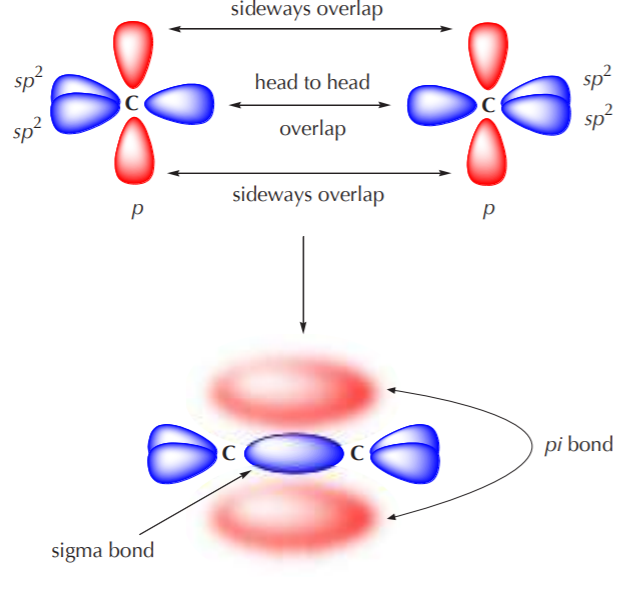
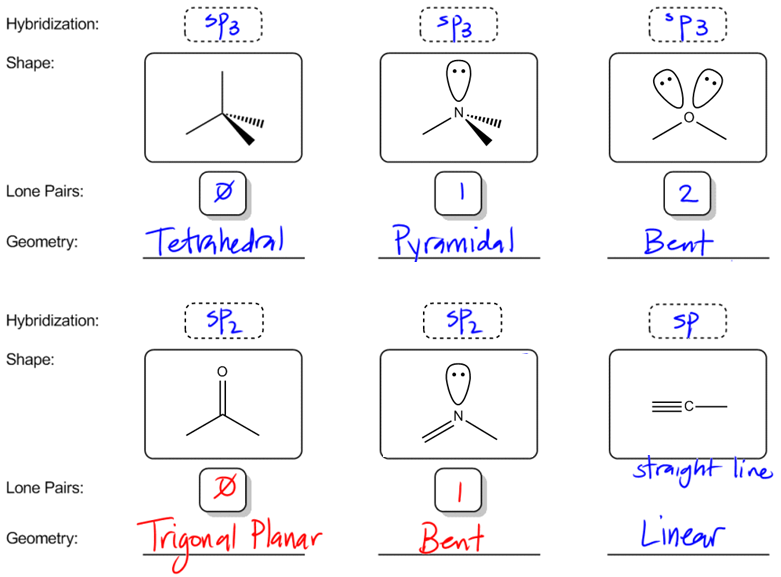
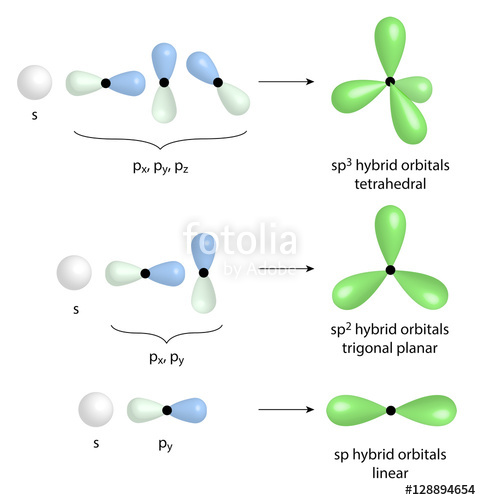
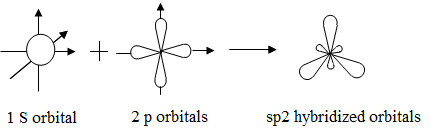
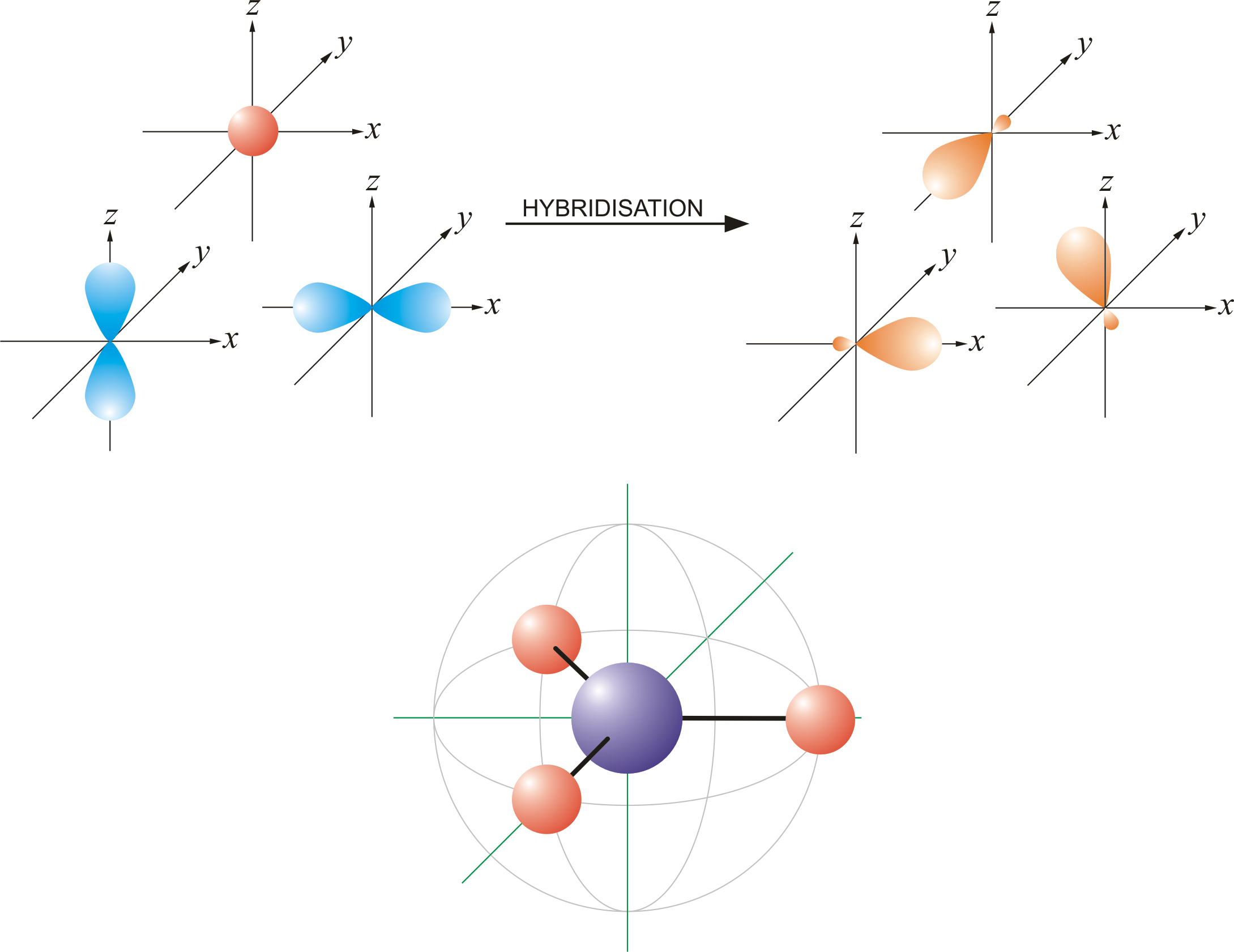
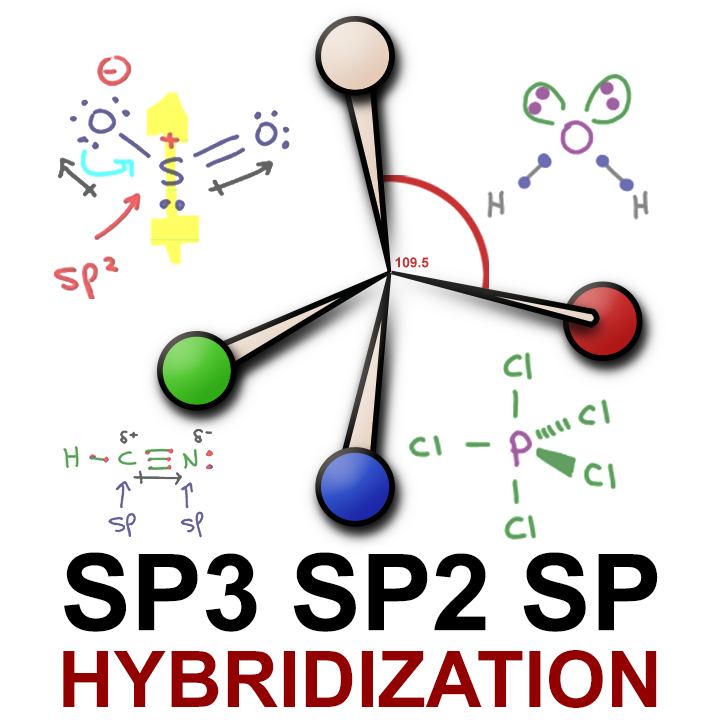
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora

A molecule has sp2 hybridization with 1 lone pair. a. The electron pair geometry of this molecule is: b. The geometry of this molecule is: c. This molecule will have an approximate
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora