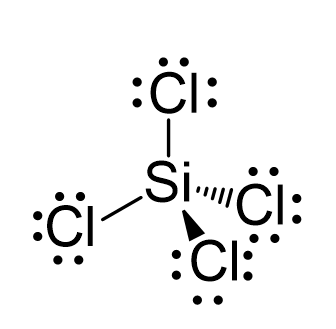
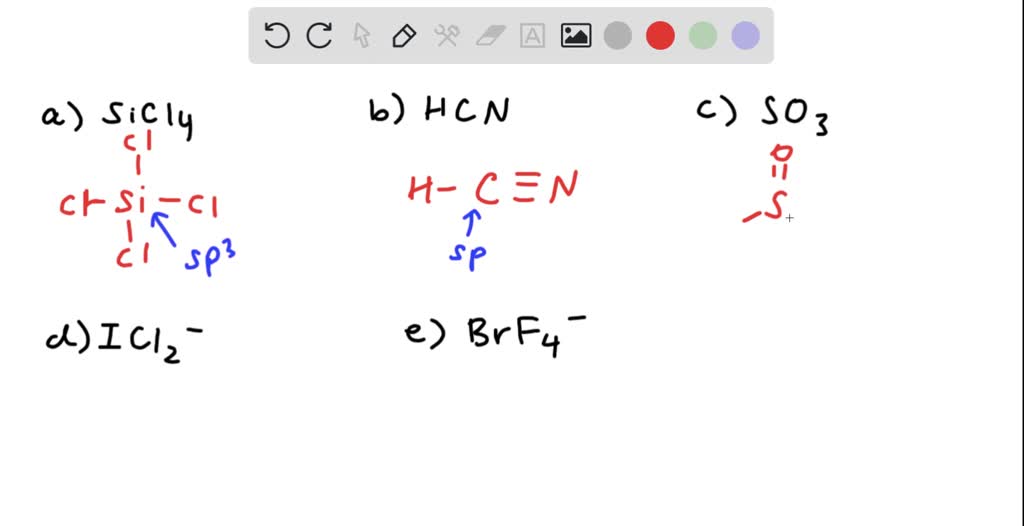
SOLVED: 4a) Using Valence Bond Theory, show the hybridization and bonding scheme for silicon tetrachloride (SiCl4): (a) write the atomic orbital diagram for the central atom, (b) circle the atomic orbitals that
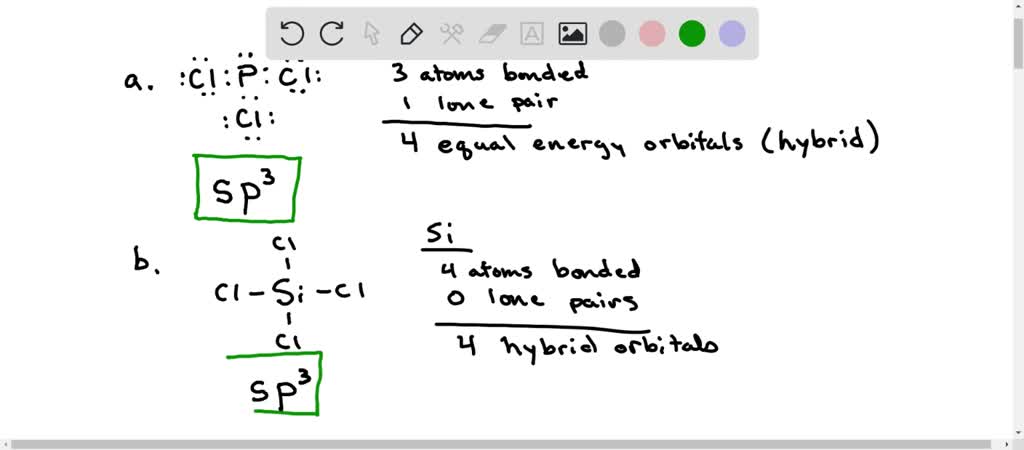
SOLVED: 10.50. What hybrid orbitals would be expected for the central atom in each of the following molecules or ions? a. PCl3 b. SiCl4 c. BeF2 d. SO2
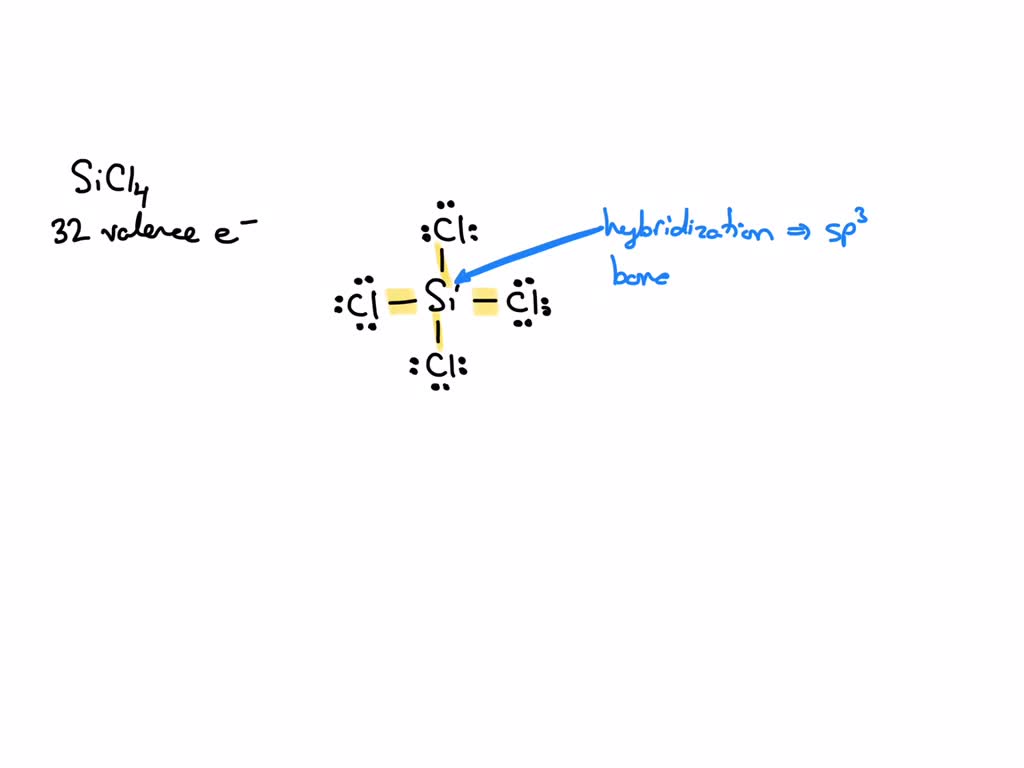
SOLVED: What is the hybridization of the central atom in SiCl4? Hybridization = What are the approximate bond angles in this substance? Bond angles = fill in the blank 2° B. What

Draw the Lewis structure for SiCl4. How many bonds are around the central atom, and what is the shape of this molecule? | Homework.Study.com
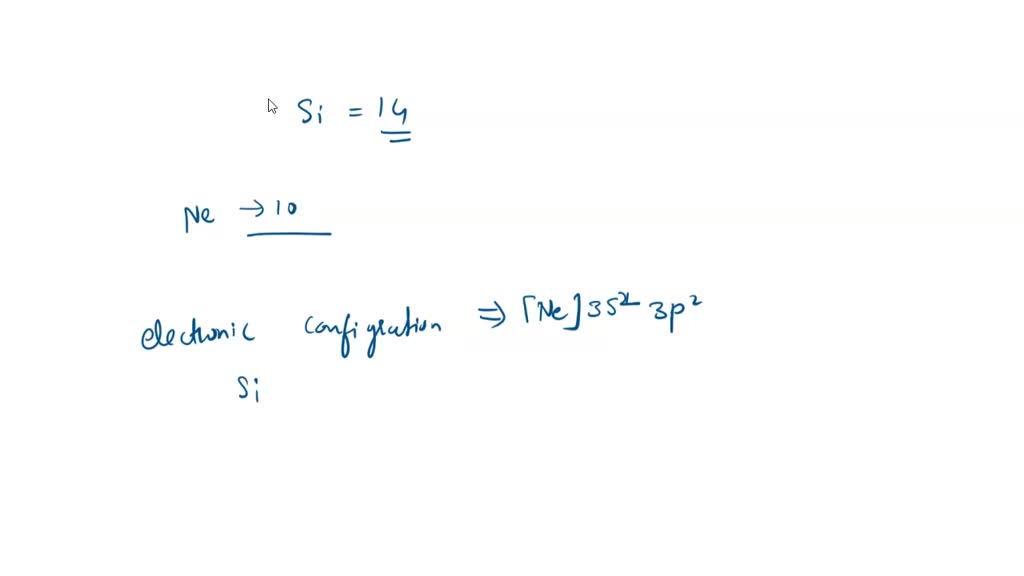
SOLVED: For the tetrahedral molecule, silicon tetrachloride, SiCl4, write the condensed electron configuration of a lone silicon atom, predict the hybridization in the molecule, and write an electron configuration for the hybridized

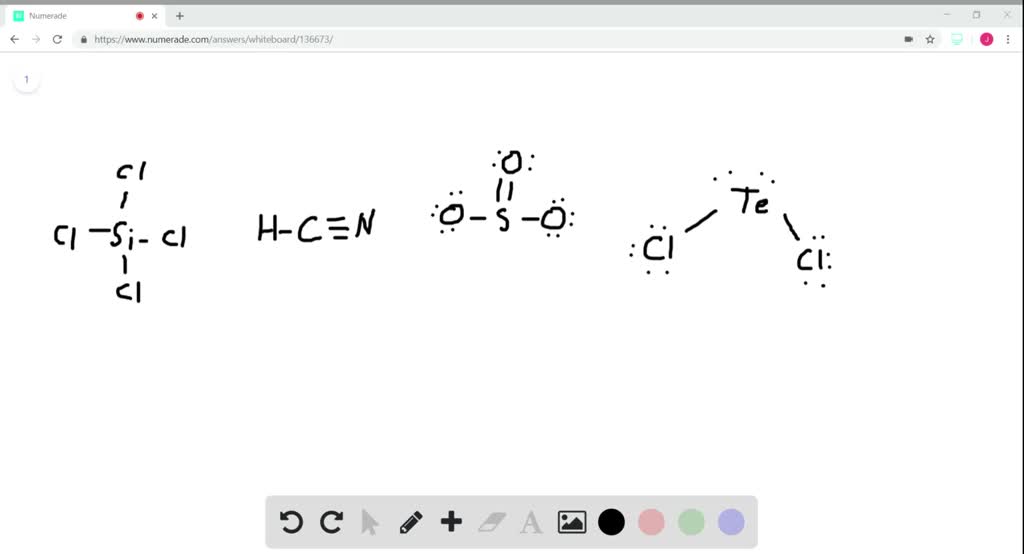
SOLVED: What is the hybridization of the central atom in each of the following: Drag the appropriate items to their respective bins. FeCl2 HCN TeCl2 SiCl4 SO2

Total number of molecules which hydrolysed at room temperature and hybridization of central atom ... - YouTube
Silicon tetrachloride SiCl4: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure –