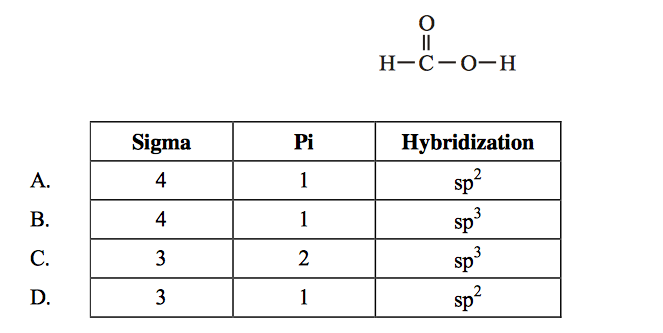
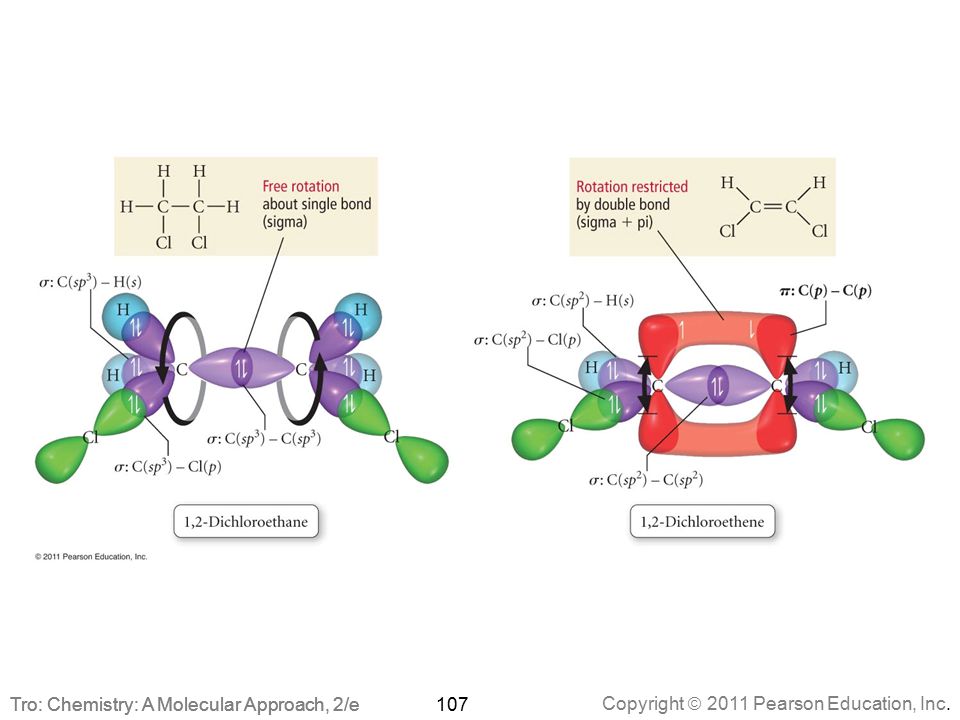
N2H2 Molecular Geometry, Bond Angles & Electron Geometry (Diimide) | Molecular geometry, Molecular, Geometry

SOLVED: What is the hybridization of each nitrogen in the N2H2 molecule? The Lewis structure is given. A-N=N-H
DFT optimized geometries of (a) *H, (b) *N2, (c) *N2H, (d) *N2H2, (e)... | Download Scientific Diagram

SOLVED: According to valence bond theory, what is the number of hybrid orbitals and the type of hybridization of the nitrogen atom in the N2H2 molecule? The Lewis structure is given as
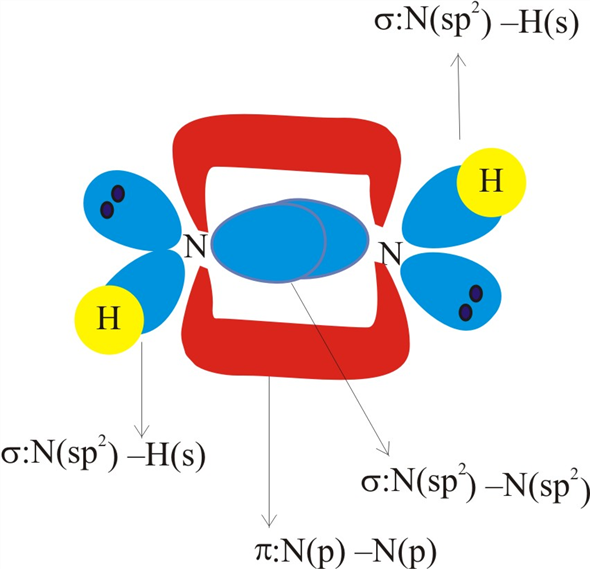


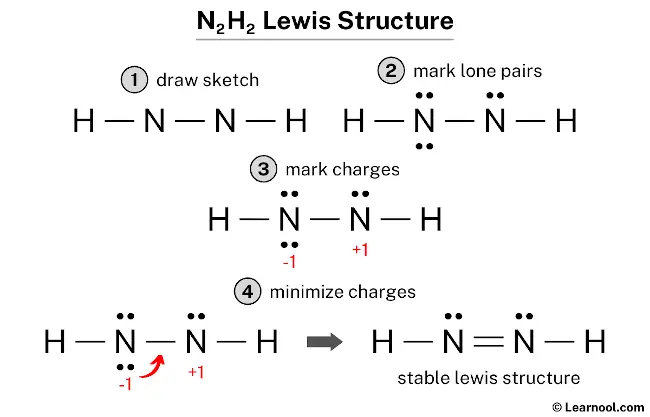










![14.2/S2.2 .16 Lewis, hybridization (sp3,sp2,sp) , shapes and angles [HL IB Chemistry] - YouTube 14.2/S2.2 .16 Lewis, hybridization (sp3,sp2,sp) , shapes and angles [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/x1fUDQKztsY/maxresdefault.jpg)