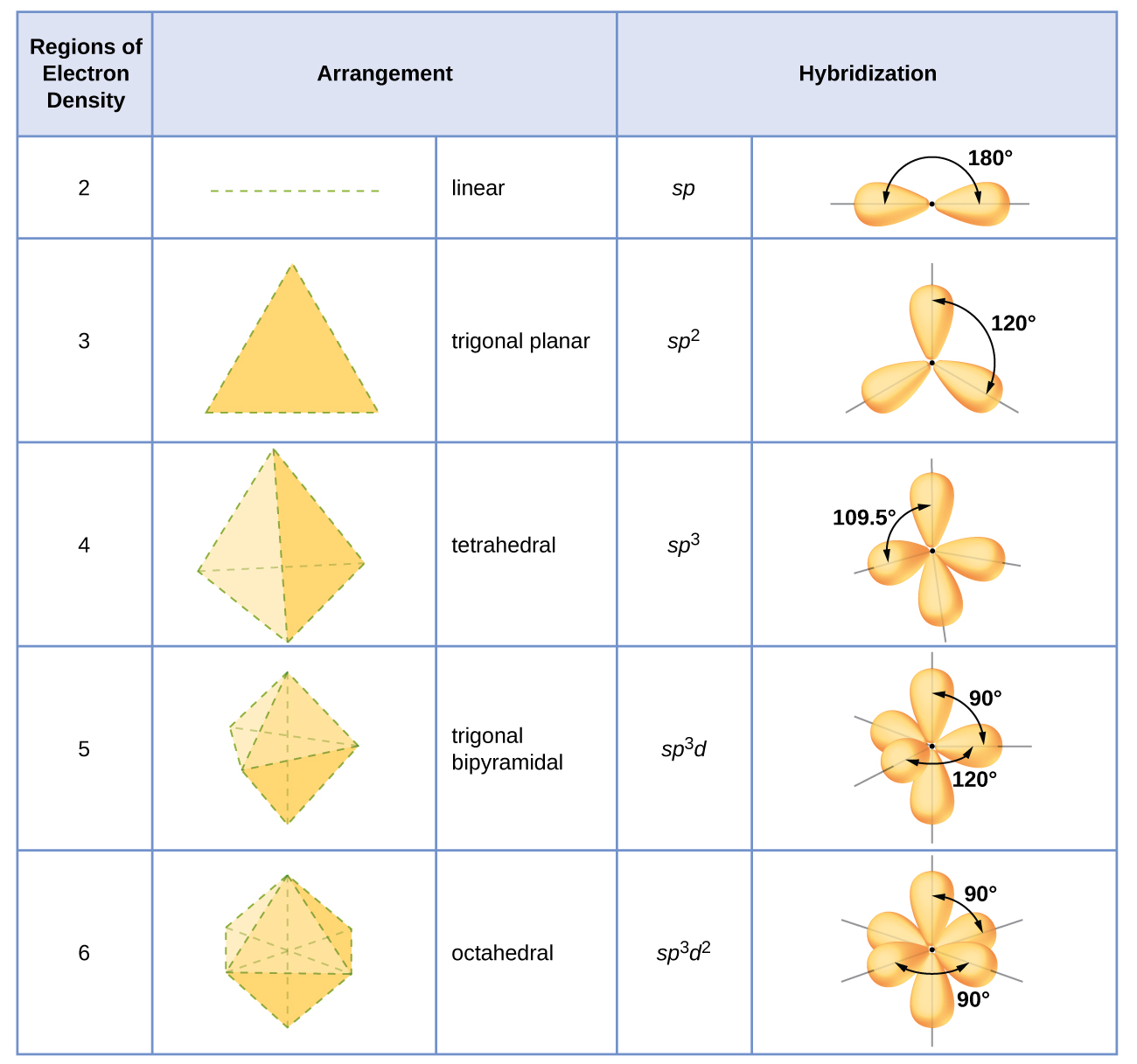
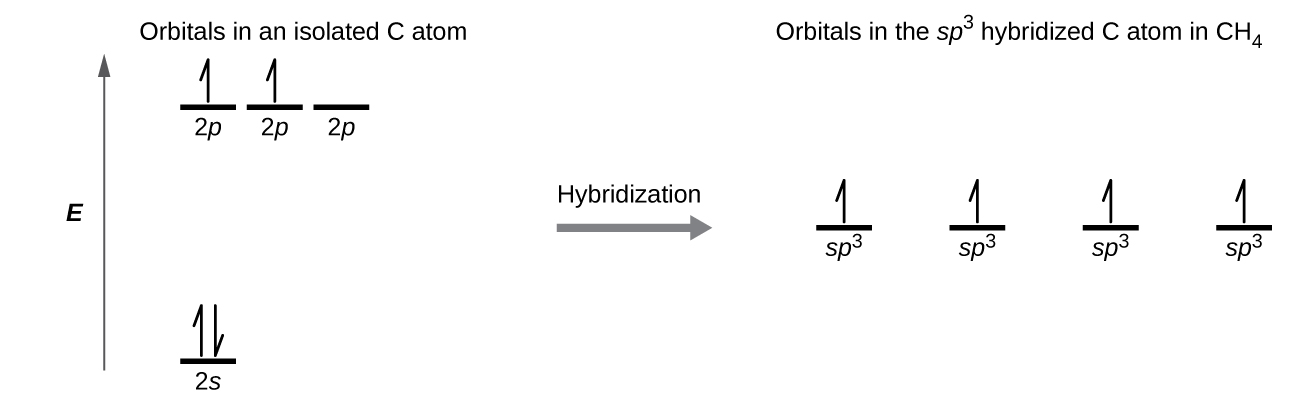
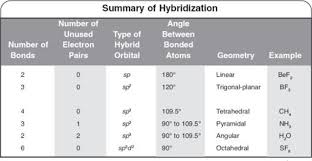
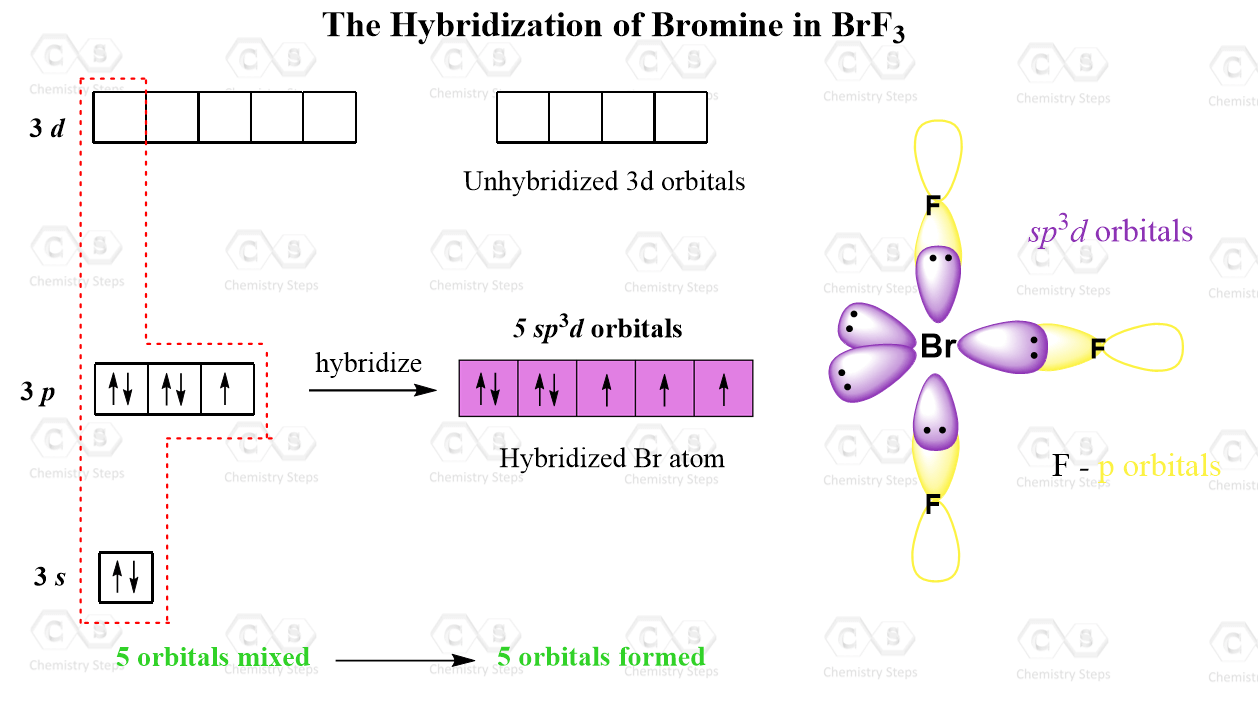
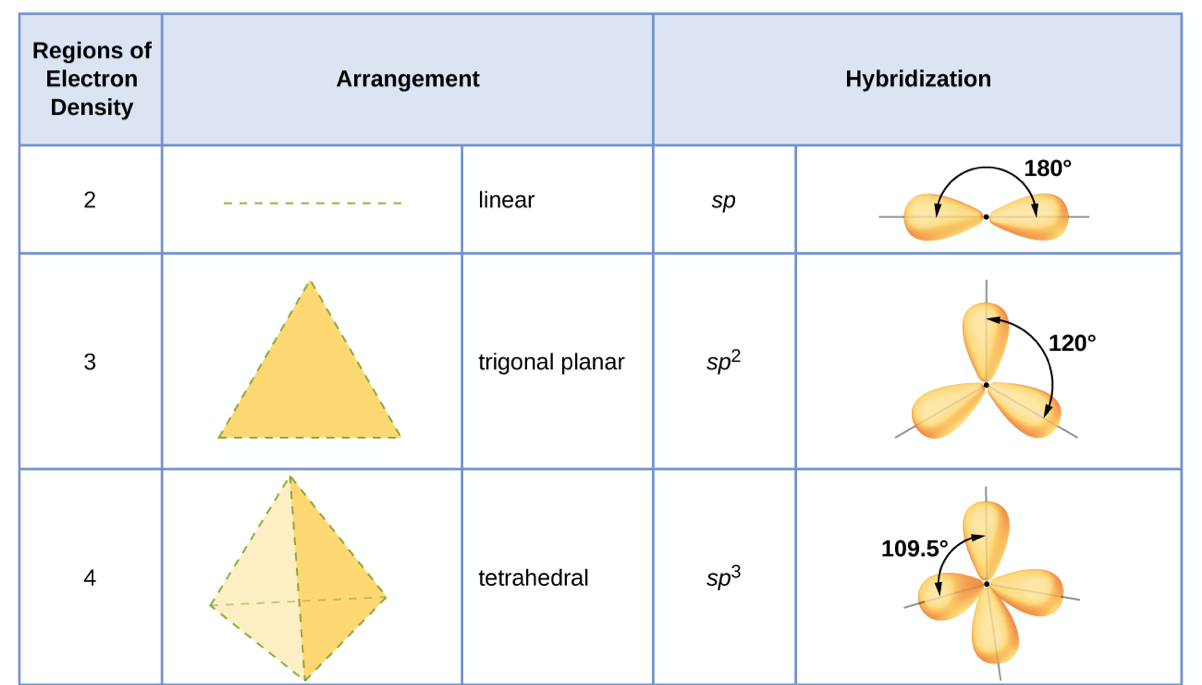
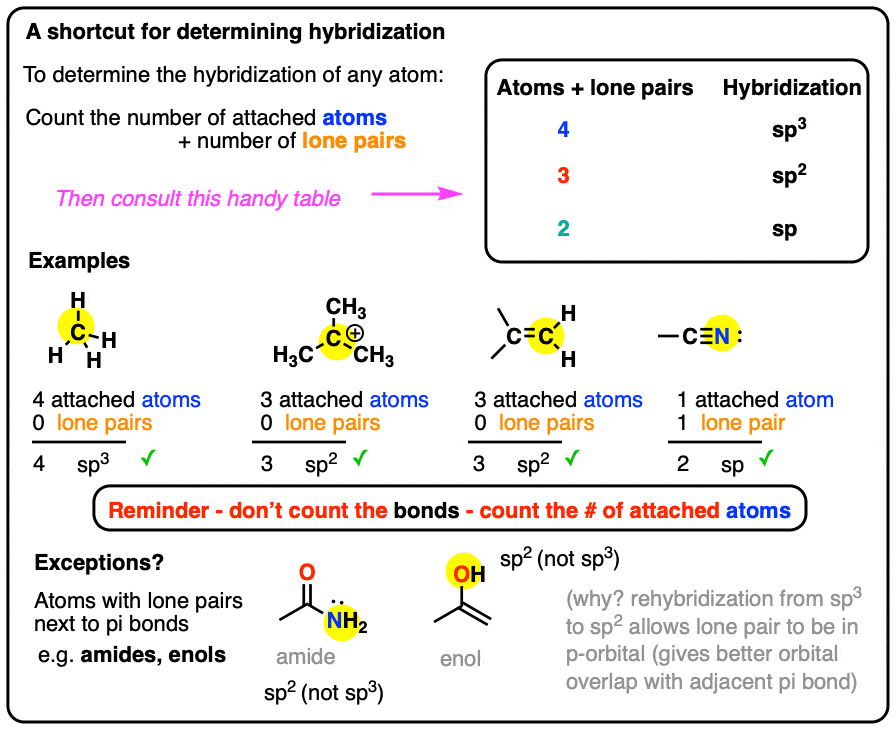
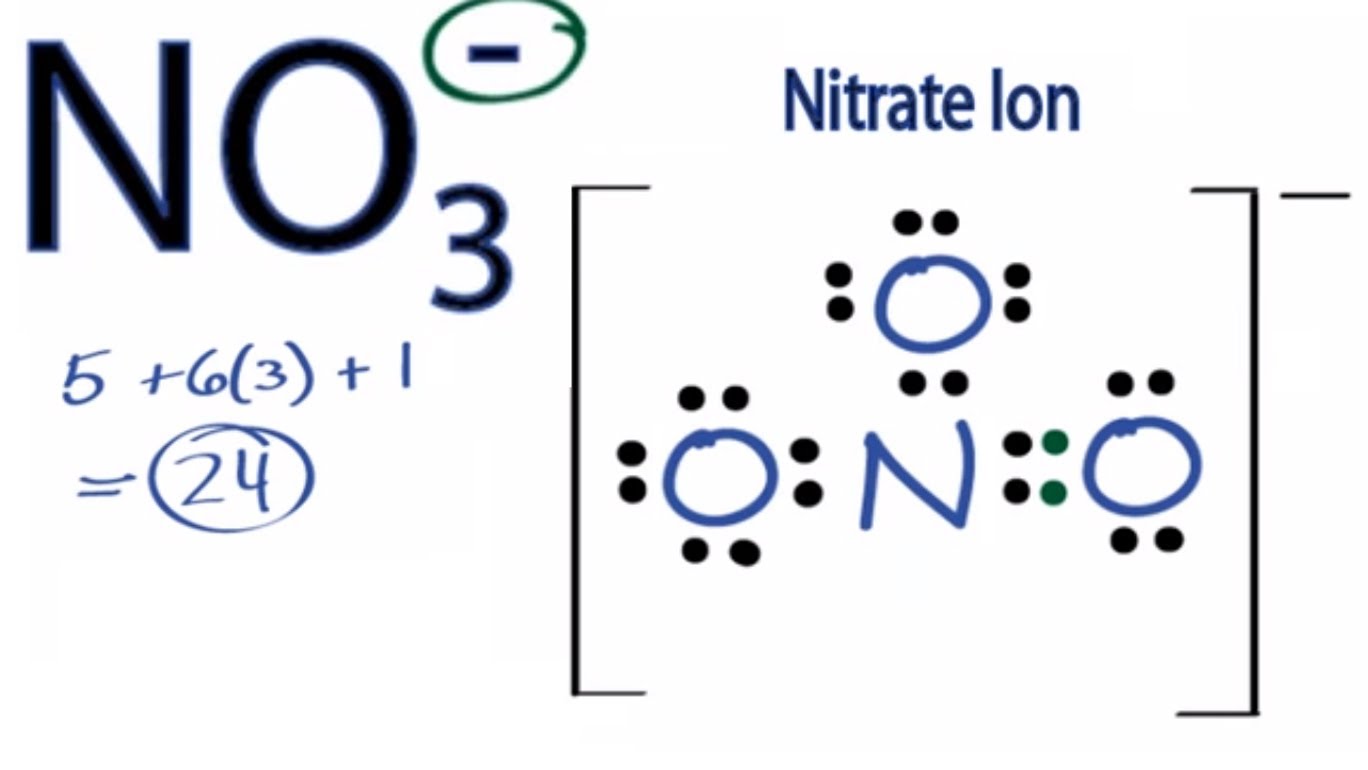
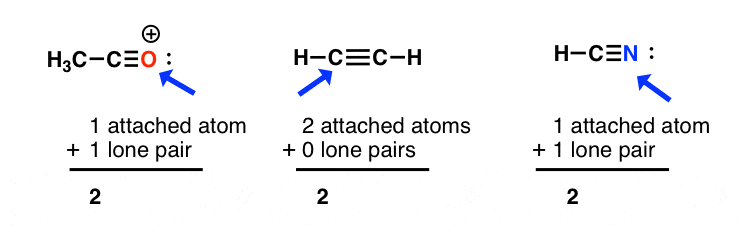
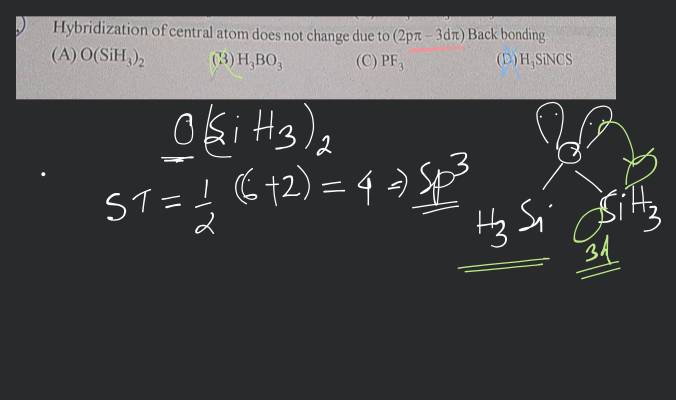In which of the following pairs of molecules/ions, the central atoms have sp^{2} hybridization?BF_{3} and NO{_{2}}^{-}NO{_{2}}^{-} and NH_{3}BF_{3} and NH{_{2}}^{-}NH{_{2}}^{-} and H_{2}O
Hybridization of central atoms in the moleculesN(CH3)3 and N(SIH3) respectively are(1) sp2 and sp2(2) sp3 and sp3(3) sp2 and sp34) sp3 and sp2
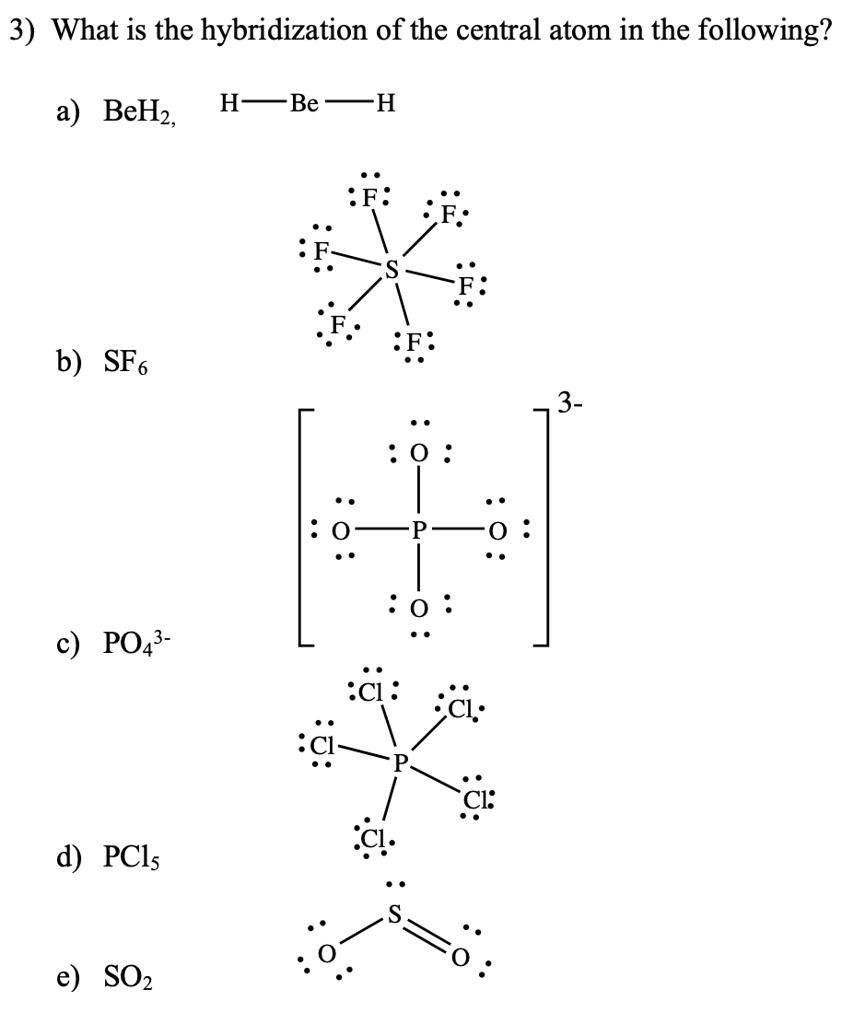
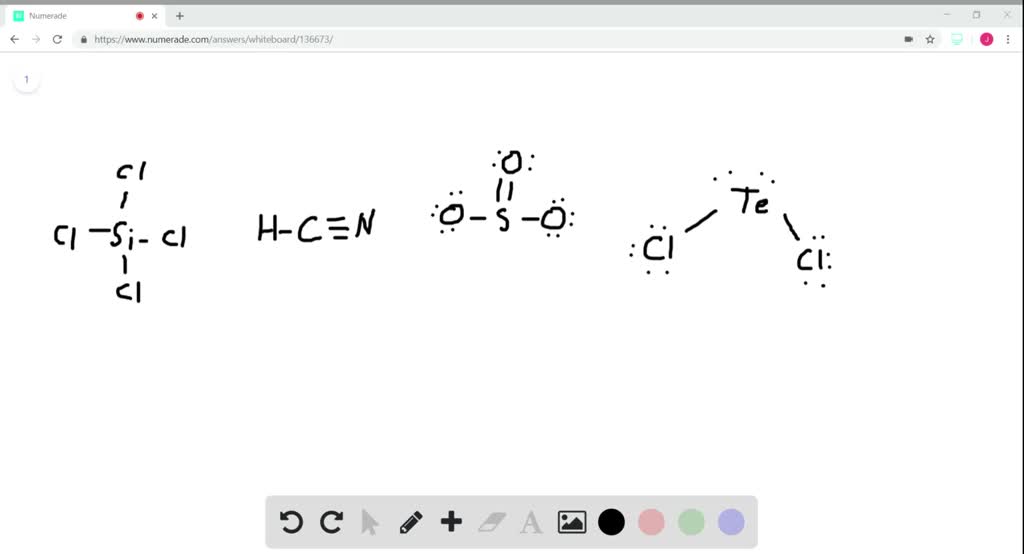
SOLVED: What is the hybridization of the central atom in the following? H2O a) BeH2, b) SF6 c) PO3^- d) PCl5 e) SO2

Specify the hybridization of the central atom in the following species respectively N 3 , NOCl , N 2 O :A. sp , sp 2, spB. sp 2, sp , spC. sp 2, sp 2, spD. sp , sp , sp 3